Introduction to Kohlrausch Law
Kohlrausch Law, commonly referred to as the Law of Independent Migration of Ions, is a foundational concept in electrochemistry. It explains how ions contribute to the electrical conductivity of electrolyte solutions, particularly under conditions of extreme dilution.
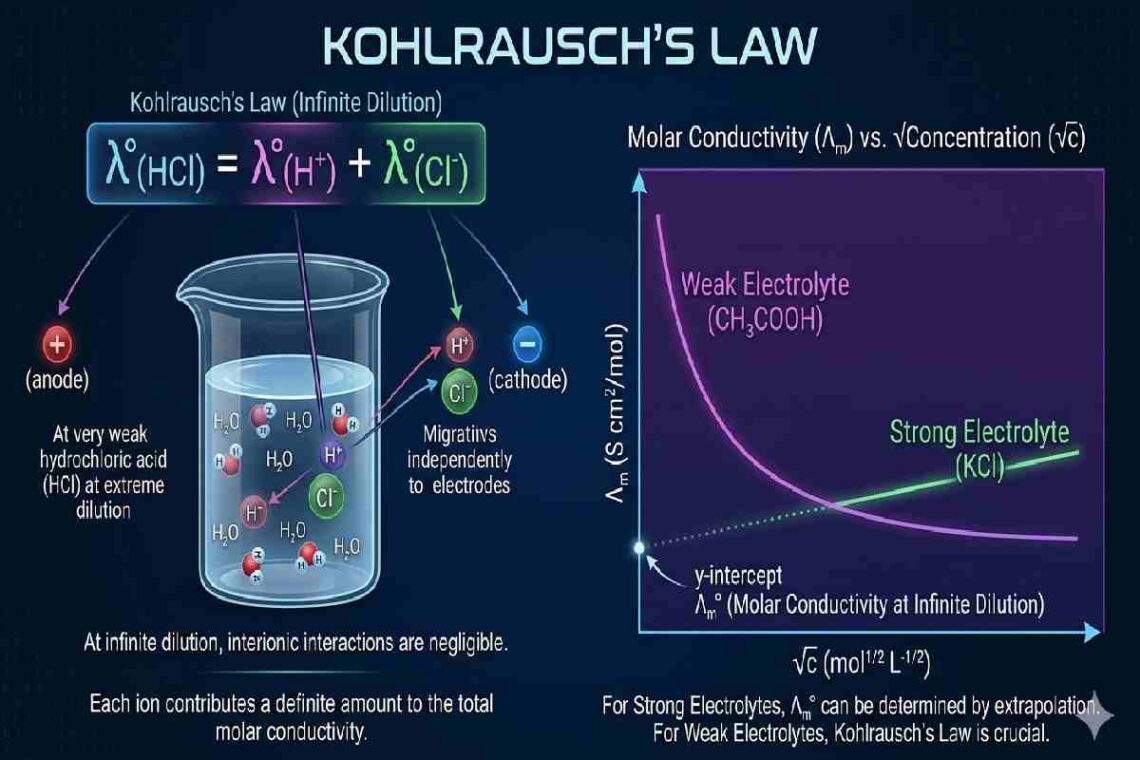
According to this law, when an electrolyte is dissolved in a solvent and diluted to an infinite extent, each ion behaves independently and contributes a fixed value to the total conductivity. This means the conductivity of the solution becomes the sum of the individual ionic conductivities.
Although the law originates in chemistry, it carries indirect but meaningful relevance in legal studies. Fields such as forensic science, environmental law, pharmaceutical regulation, and industrial compliance rely on chemical analysis, where conductivity measurements play a role. For law students and professionals, understanding such scientific principles strengthens the interpretation of technical evidence and expert testimony.
Historical Background and Origin
Kohlrausch’s Law was developed in the late nineteenth century by the German physicist Friedrich Kohlrausch. His experimental work on the conductivity of electrolyte solutions marked a turning point in electrochemical research.
Kohlrausch’s experiments demonstrated that ions possess inherent conductive properties that remain constant at infinite dilution. His findings were later validated and expanded upon by other scientists, including Svante Arrhenius, who introduced the theory of electrolytic dissociation.
Key Historical Developments
| Period | Contribution |
| 1870s | Experimental studies on ionic conductivity |
| 1875 | Formal statement of Kohlrausch Law |
| Late 19th century | Empirical validation of ionic independence |
| Early 20th century | Integration with dissociation theory |
This historical progression highlights how Kohlrausch Law became a cornerstone of modern electrochemistry.
Scientific Principles Behind Kohlrausch Law
Core Statement of the Law
Kohlrausch Law states:
At infinite dilution, the molar conductivity of an electrolyte is equal to the sum of the conductivities of its individual ions.
Where:
- : Molar conductivity at infinite dilution
- : Ionic conductivity of the cation
- : Ionic conductivity of the anion
Fundamental Concepts
Electrolytes
Electrolytes are substances that dissociate into ions when dissolved in a solvent such as water. They are classified into:
- Strong electrolytes: Completely dissociate (e.g., NaCl, HCl)
- Weak electrolytes: Partially dissociate (e.g., acetic acid)
Conductivity
Conductivity refers to the ability of a solution to carry electric current. This depends on:
- Number of ions present
- Charge on ions
- Mobility of ions
Infinite Dilution
Infinite dilution is a theoretical condition where:
- Ion–ion interactions become negligible
- Each ion moves independently
- Maximum conductivity is achieved
Interpretation of Ionic Independence
At high dilution levels, the forces of attraction between oppositely charged ions decrease significantly. As a result:
- Ions migrate freely under an electric field
- Their contribution to conductivity becomes additive
- The total conductivity becomes predictable
Illustrative Table of Ionic Contributions
| Electrolyte | Expression at Infinite Dilution |
| NaCl | λ(Na⁺) + λ(Cl⁻) |
| HCl | λ(H⁺) + λ(Cl⁻) |
| KNO₃ | λ(K⁺) + λ(NO₃⁻) |
This additive property allows scientists to calculate unknown ionic conductivities using known values.
Assumptions of Kohlrausch Law
| Assumption | Explanation |
| Independent ion movement | No interionic forces at infinite dilution |
| Complete dissociation | Electrolytes fully ionize |
| Constant temperature | Conductivity depends on thermal stability |
| No ion pairing | Ions do not recombine in solution |
These assumptions are critical for the validity of the law and define its scope of application.
Applications of Kohlrausch Law
Kohlrausch Law has broad applications in scientific analysis and practical fields, some of which intersect with legal frameworks.
Determination of Molar Conductivity of Weak Electrolytes
Weak electrolytes do not fully dissociate, making direct conductivity measurement challenging. Kohlrausch’s Law allows indirect calculation:
This method is widely used in analytical chemistry laboratories.
Degree of Dissociation
The law helps determine the extent to which a weak electrolyte dissociates:
Where:
- : Degree of dissociation
- : Molar conductivity at given concentration
Solubility of Sparingly Soluble Salts
Kohlrausch Law can be used to determine solubility by measuring conductivity in saturated solutions.
Forensic and Legal Relevance
In legal contexts, particularly forensic investigations:
- Conductivity measurements help identify unknown substances
- Chemical evidence can be validated through ionic analysis
- Laboratory reports often rely on principles derived from Kohlrausch Law
This is particularly relevant in:
- Drug analysis cases
- Toxicology reports
- Environmental contamination disputes
Industrial and Environmental Monitoring
Industries use conductivity measurements to:
- Monitor water purity
- Ensure chemical compliance
- Maintain safety standards
Regulatory bodies in India often rely on such measurements for environmental assessments.
Data Comparison of Electrolytes
| Electrolyte Type | Conductivity Behavior | Example | Legal Relevance |
| Strong electrolyte | High conductivity | NaCl | Industrial compliance |
| Weak electrolyte | Low conductivity | CH₃COOH | Pharmaceutical testing |
| Non-electrolyte | No conductivity | Glucose | Food regulation |
Relevance of Kohlrausch Law in the Indian Context
While Kohlrausch Law is primarily a scientific principle, its implications extend into multiple domains regulated by Indian law. Legal professionals increasingly encounter scientific data in litigation, compliance, and regulatory matters, making foundational knowledge of such principles valuable.
Environmental Law and Water Quality Regulation
In India, environmental protection is governed by statutes such as the Water (Prevention and Control of Pollution) Act, 1974 and monitored by bodies like the Central Pollution Control Board.
Conductivity measurements—based on principles like Kohlrausch Law—are routinely used to:
- Assess water pollution levels
- Detect dissolved ionic contaminants
- Monitor industrial discharge
High conductivity in water often indicates excessive ionic content, which may signal contamination by industrial effluents. Legal disputes involving pollution frequently rely on laboratory data derived from conductivity analysis.
Forensic Science and Evidence Law
Under the Indian Evidence Act, scientific evidence plays a crucial role in judicial proceedings. Forensic laboratories utilize conductivity measurements to analyze:
- Unknown chemical substances
- Toxic compounds
- Drug samples
Kohlrausch’s Law indirectly supports these analyses by enabling the accurate determination of ionic properties in dilute solutions.
Expert testimony based on such chemical analysis must meet standards of reliability and scientific validity, making an understanding of underlying principles essential for legal practitioners.
Pharmaceutical and Drug Regulation
India’s pharmaceutical sector is regulated under the Drugs and Cosmetics Act, 1940. Quality control laboratories employ conductivity measurements to:
- Verify chemical composition
- Ensure purity of drugs
- Detect adulteration
Weak electrolyte analysis—enabled by the Kohlrausch Law—is particularly important in drug formulation and testing. Legal issues such as patent disputes, quality violations, and regulatory compliance often involve such data.
Industrial Compliance and Standards
Industries must adhere to environmental and safety standards set by authorities such as the Bureau of Indian Standards.
Conductivity testing is used in:
- Water treatment plants
- Chemical manufacturing units
- Food processing industries
Non-compliance can lead to:
- Legal penalties
- Closure orders
- Environmental litigation
Thus, the Kohlrausch Law underpins analytical methods that influence regulatory enforcement.
Advanced Applications and Interpretations
Beyond basic applications, the Kohlrausch Law supports several advanced analytical techniques.
Ionic Mobility and Transport Numbers
The law helps determine ionic mobility, which refers to the speed of an ion under an electric field. This is crucial in:
- Electrochemical cell design
- Battery technology
- Industrial electrolysis
Electrochemical Research
Modern electrochemistry builds upon Kohlrausch’s findings to study:
- Ion transport mechanisms
- Conductance behavior at varying concentrations
- Interaction effects at non-ideal conditions
Analytical Chemistry Techniques
Kohlrausch Law is applied in:
- Conductometric titrations
- Determination of dissociation constants
- Study of equilibrium systems
These techniques are widely used in laboratories that support legal investigations and industrial compliance.
Comparative Analysis of Kohlrausch Law Applications
| Field | Application | Practical Outcome | Legal Impact |
| Environmental science | Water conductivity testing | Pollution detection | Regulatory enforcement |
| Forensic science | Substance identification | Evidence validation | Court admissibility |
| Pharmaceuticals | Drug purity testing | Quality assurance | Compliance and litigation |
| Industry | Process monitoring | Safety and efficiency | Legal compliance |
Limitations of Kohlrausch Law
Despite its importance, Kohlrausch Law has certain limitations that must be acknowledged.
Dependence on Infinite Dilution
The law strictly applies only at infinite dilution. At higher concentrations:
- Ion interactions increase
- Conductivity deviates from predicted values
Temperature Sensitivity
Conductivity varies with temperature. Accurate application requires controlled conditions.
Not Applicable to Strong Interactions
In solutions where:
- Ion pairing occurs
- Strong electrostatic forces exist
The assumptions of the law break down.
Practical Examples
Example: Calculation of Ionic Conductivity
Using known values:
| Electrolyte | Molar Conductivity (S cm² mol⁻¹) |
| HCl | 426 |
| NaCl | 126 |
| CH₃COONa | 91 |
We can calculate:
This demonstrates how unknown values are derived using known ionic contributions.
Example: Environmental Testing
In a legal case involving water contamination:
- Conductivity readings are taken
- High ionic concentration is detected
- Source traced to industrial discharge
Such evidence may be used in court to establish liability.
Conclusion
Kohlrausch Law remains a cornerstone of electrochemistry, providing critical insight into the behavior of ions in dilute solutions. Its principle—that ions contribute independently to conductivity at infinite dilution—has enabled advancements in analytical chemistry, industrial processes, and scientific research.
For law students and legal professionals, the relevance of Kohlrausch Law lies in its application within regulated environments. Whether in environmental litigation, forensic analysis, or pharmaceutical compliance, the law supports the interpretation of scientific data that frequently appears in legal contexts.
In India, where regulatory frameworks increasingly rely on scientific evidence, interdisciplinary understanding is essential. Kohlrausch Law exemplifies how a scientific concept can have far-reaching implications beyond its original domain, influencing legal reasoning, policy enforcement, and judicial outcomes.